Research and Innovation are central to the vision for TU Dublin as a technological university. As the world faces significant challenges relating to, amongst others, climate change, biodiversity loss, inequality, political instability, public health, our university is focussing on, as outlined in our Strategic Intent 2030, the “creation of new knowledge and the development of timely & practical solutions that address the SDGs.”
Our collective commitment to working towards the UN Sustainable Development Goals is consistent with our longstanding culture of focussing research and innovation activity on delivery of positive impact in our society. Solutions to the SDGs require concerted efforts across society, with universities playing a key role in providing open and creative environments in which new ideas can be explored and tested.
The TU Dublin community has the energy and drive to play our part. Whilst we will actively support research and innovation activity from across the broad spectrum of disciplines pursued across all five TU Dublin faculties, we identify in this strategy a set of five research priorities, aligned to selected SDGs, where we believe that coalescing our efforts in a transdisciplinary manner will focus and amplify the excellence and impact of our work.

Priority Areas & Expertise
Our researchers provide deep insights and solutions that are transforming and sustaining society through knowledge creation, underpinned by novel theoretical concepts and often with a practical and practice-based approach.
Read More

Impact
We are proud to say that in TU Dublin our research outputs make a significant contribution to new knowledge, products, processes, and thinking.
Read More

Collaborations
We have strong collaborative research relationships with partners in business, industry, with government and non-government organisations and communities. We have experienced teams to match your needs to our research expertise and facilities.
Read More

Facilities
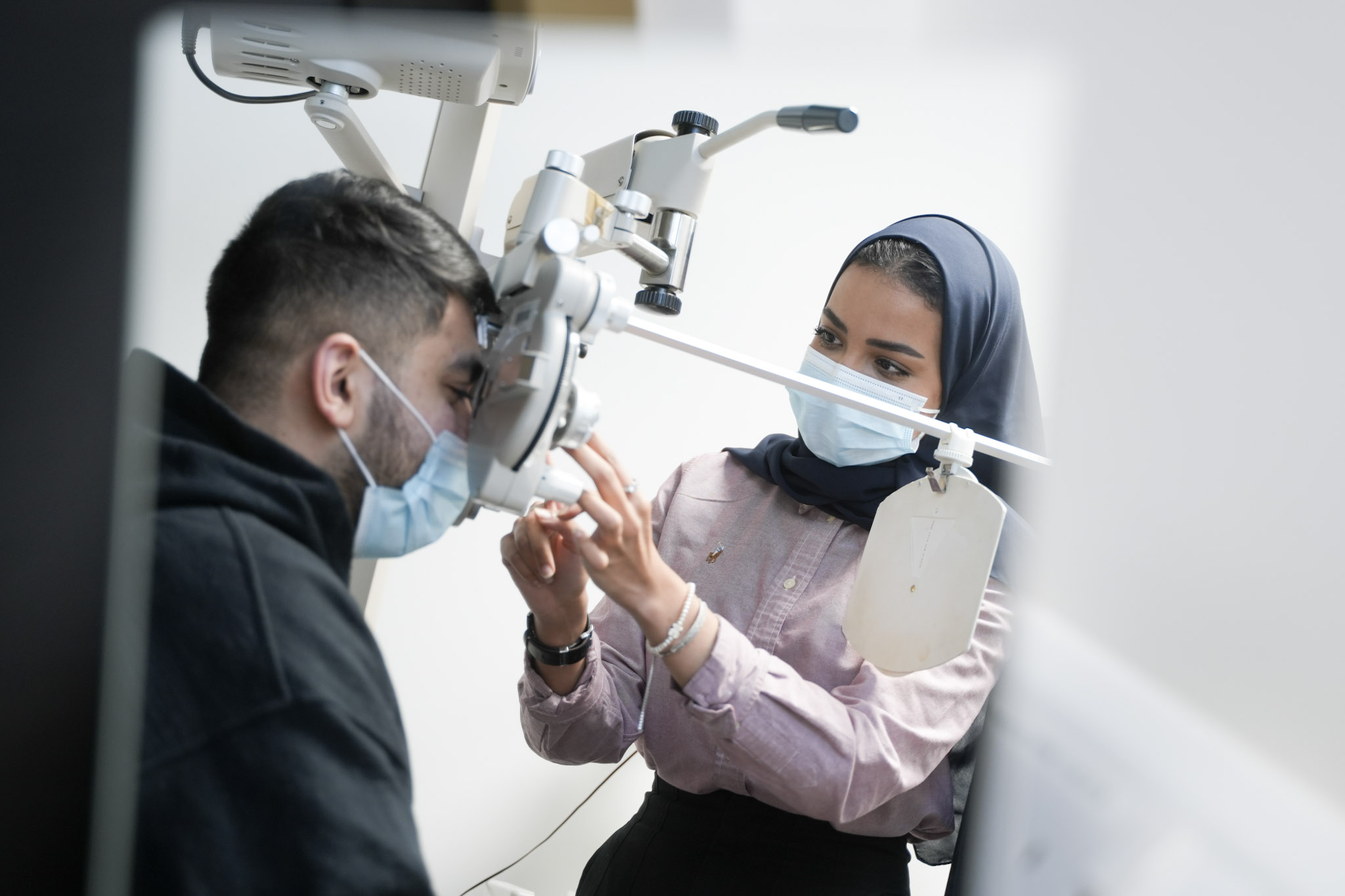
Our experts deliver trusted results, generate new knowledge, & develop groundbreaking solutions to real-world challenges in state-of-the-art research facilities & laboratories specialised equipment. We facilitate collaborative research projects, experiments, data capture & analysis.
Read More

Postgraduate Research
Our research degree programmes provide you with the opportunity to work with leading research academics in addressing some of the worlds most pressing challenges. You will become part of a vibrant community & develop your research skills in a dynamic supportive environment.
Read More

Support for Researchers
The R&I team provides dedicated, specialist support to help researchers develop proposals, manage projects, disseminate their findings, and bring their research to a commercial or societal application.
Read More
What's New?
Impact Case Studies
Research and Innovation at a Glance
400+
Research Projects
20,000+
Citations
400+
Sustainable Businesses
€700m
Economic Value